|
In single covalent bonds, typically both atoms in the bond. Valence electrons are outer shell electrons with an atom and can participate in the formation of chemical bonds. Because much of the chemistry of an element is influenced by valence electrons, we would expect that these elements would have similar chemistry- and they do. This page titled 4: Valence Electrons and Bonding is shared under a CC BY-NC-SA 4.0 license and was authored, remixed, and/or curated by Elizabeth Gordon. They all have a similar electron configuration in their valence shells: a single s electron. How to determine the number of valence electrons and draw Lewis structures for main group elements starting from the electron configuration. Look at the group that the element is in, as the group number. How many valence electrons does an element have You can use the periodic table to help you determine how many valence electrons an element (specifically, a neutral atom of the element) has. Their electron configurations (abbreviated for the larger atoms) are as follows, with the valence shell electron configuration highlighted: Table shows first column of the periodic table and their electron configurations. On the other hand, nitrogen can form NH 3 so it has a valence of 3, and 3 valence electrons. 
For example, take the elements in the first column of the periodic table: H, Li, Na, K, Rb, and Cs. 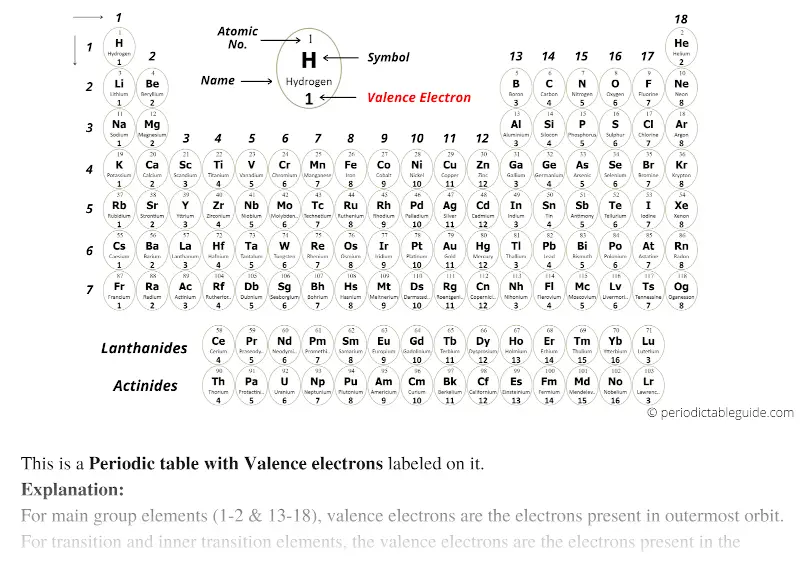
If we look at just the valence shell's electron configuration, we find that in each column, the valence shell's electron configuration is the same. (The inner electrons are called core electrons.) The valence electrons largely control the chemistry of an atom. The electrons in the highest-numbered shell, plus any electrons in the last unfilled subshell, are called valence electrons the highest-numbered shell is called the valence shell. Atoms share electrons and form covalent bonds to satisfy the octet rule. 4.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. The atoms in group 6A make two covalent bonds. 
The periodic table is separated into blocks depending on which subshell is being filled for the atoms that belong in that section. Covalent bonds are formed by two atoms sharing electrons.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
